Snail Fever Vaccine Reaches First Human Clinical Trial
Long journey to prevent major tropical disease is worth the wait
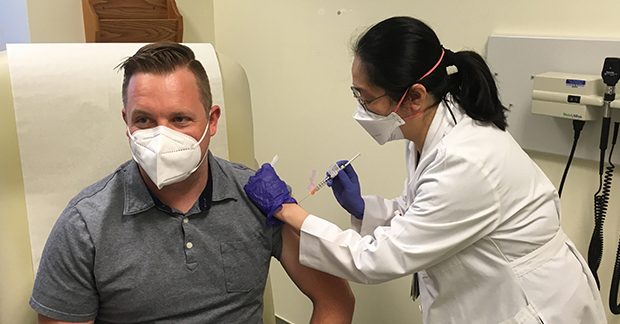
Participant in clinical trial receives world’s first-ever injection of SchistoShield
After decades of hard work, a preventative vaccine for major tropical disease schistosomiasis has reached a Phase 1 clinical trial, providing a dose for the first healthy human volunteer and paving the way into a better future for the 74 countries and nearly one billion people currently threatened by this disease.
Afzal A. Siddiqui, Ph.D., Grover E. Murray Distinguished Professor with Texas Tech University Health Sciences Center (TTUHSC) School of Medicine, served as lead researcher and invented the schistosomiasis vaccine, called SchistoShield®. This vaccine was brought about by PAI Life Sciences, a biotechnology company specializing in translational research for neglected tropical diseases.
According to Siddiqui, it is the result of many years of hard work and research.

Afzal A. Siddiqui, Ph.D.
“It has been 30 years of a bumpy ride at a snail’s pace to bring SchistoShield® vaccine for snail fever [schistosomiasis] to first in human clinical trials,” said Siddiqui.
Schistosomiasis (or ‘snail fever’) is a disease caused by parasitic flatworms predominantly found in sub-Saharan Africa, Middle East, Southeast Asia and the Caribbean. These worms reach humans through the skin when it comes into contact with contaminated water. Symptoms of schistosomiasis include abdominal pain, diarrhea and blood in stools and/or urine. With the potential to cause long-term liver damage, kidney failure and death, a clinically proven vaccine could change the course of the future for millions.
“Development of a vaccine that is effective against geographically distinct forms of schistosomiasis is essential to make a meaningful impact in global reduction of disease burden,” said Siddiqui. “A low-cost, effective vaccine for schistosomiasis would greatly aid in the fight against this debilitating disease. The significance of successful clinical trials is clear in that an approved, effective, deployable vaccine for this disease would impact as many as one billion people in 74 countries throughout the world.”
In May of this year, TTUHSC achieved Carnegie Classification® status for Special Focus Four-Year Research Institutions.
The clinical trial will evaluate the safety and the immunogenicity of PAI Life Sciences’s SchistoShield® vaccine in 45 healthy adults between the ages of 18 and 55, and is sponsored by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health.
Related Stories
TTUHSC Experts Receive Junior Investigator Research Grants through Texas Alzheimer’s Research and Care Consortium
Texas Tech University Health Sciences Center (TTUHSC) researchers Aric Logsdon, Ph.D., and Jonathan Singer, Ph.D., received Junior Investigator Research Grants from the Texas Alzheimer’s Research and Care Consortium (TARCC).
CPRIT Awards TTUHSC Researcher $1.4 Million Cancer Grant in Efforts to Expand Rural Clinical Trials
The Cancer Prevention and Research Institute of Texas (CPRIT) awarded a $1.4 million grant to Erin Barr, M.D., a clinician of pediatric hematology/oncology and researcher in the School of Medicine at Texas TTUHSC. Barr’s research, “Enhancing access to pediatric oncology clinical trials in West Texas,” received funding under the Recruitment or Retention of Clinical Trialists Award.
TAMEST Executive Director Visits Texas Tech University Health Sciences Center to Strengthen Research Collaboration
TTUHSC welcomed Terrence Henry, executive director of the Texas Academy of Medicine, Engineering, Science and Technology (TAMEST), for a campus visit aimed at strengthening partnerships and expanding research engagement opportunities across Texas.
Recent Stories
TTUHSC Experts Receive Junior Investigator Research Grants through Texas Alzheimer’s Research and Care Consortium
Texas Tech University Health Sciences Center (TTUHSC) researchers Aric Logsdon, Ph.D., and Jonathan Singer, Ph.D., received Junior Investigator Research Grants from the Texas Alzheimer’s Research and Care Consortium (TARCC).
CPRIT Awards TTUHSC Researcher $1.4 Million Cancer Grant in Efforts to Expand Rural Clinical Trials
The Cancer Prevention and Research Institute of Texas (CPRIT) awarded a $1.4 million grant to Erin Barr, M.D., a clinician of pediatric hematology/oncology and researcher in the School of Medicine at Texas TTUHSC. Barr’s research, “Enhancing access to pediatric oncology clinical trials in West Texas,” received funding under the Recruitment or Retention of Clinical Trialists Award.
Texas Tech University Health Sciences Center’s Klein Selected to Leadership Role by the Texas Pharmacy Association
The Texas Pharmacy Association announced (May 19) the appointment of Mary Klein, Pharm.D., as assistant vice president of Strategic Initiatives and Practice Innovation.
